Basic Information about Mercuric Chloride (HgCl₂): The Ultimate Guide
Mercuric chloride (HgCl₂), also known as mercury(II) chloride or corrosive sublimate, is a white crystalline chemical compound with a complex legacy across science, industry, and (historically) medicine. Its high reactivity and solubility have made it invaluable for specialized applications, but its severe toxicity and environmental impact demand careful handling and strict regulatory oversight.
This comprehensive guide covers mercuric chloride’s structure, physical and chemical properties, history, uses, hazards, regulations, and best practices for safe procurement and handling in South Africa.
Table of Contents
- Introduction to Mercuric Chloride
- Chemical Structure and Properties
- Physical Characteristics
- Historical Background
- Synthesis and Industrial Production
- Laboratory and Industrial Uses
- Historical and Obsolete Medical Applications
- Safety, Toxicity, and Exposure Risks
- Environmental Impact
- Handling, Storage, and Disposal
- Regulatory Status in South Africa
- How to Purchase Safely from Brand Chemical Supplies
- Frequently Asked Questions
- References and Further Reading
- Conclusion
1. Introduction to Mercuric Chloride
Mercuric chloride (HgCl₂) is an inorganic mercury salt with the mercury atom in the +2 oxidation state. It is recognized for its:
- High chemical reactivity
- Extreme toxicity
- Historical importance in chemistry and medicine
- Current value in select laboratory and industrial applications
Despite the dangers, its unique properties mean it remains in use where no suitable alternatives exist—making knowledge and compliance essential for all users.
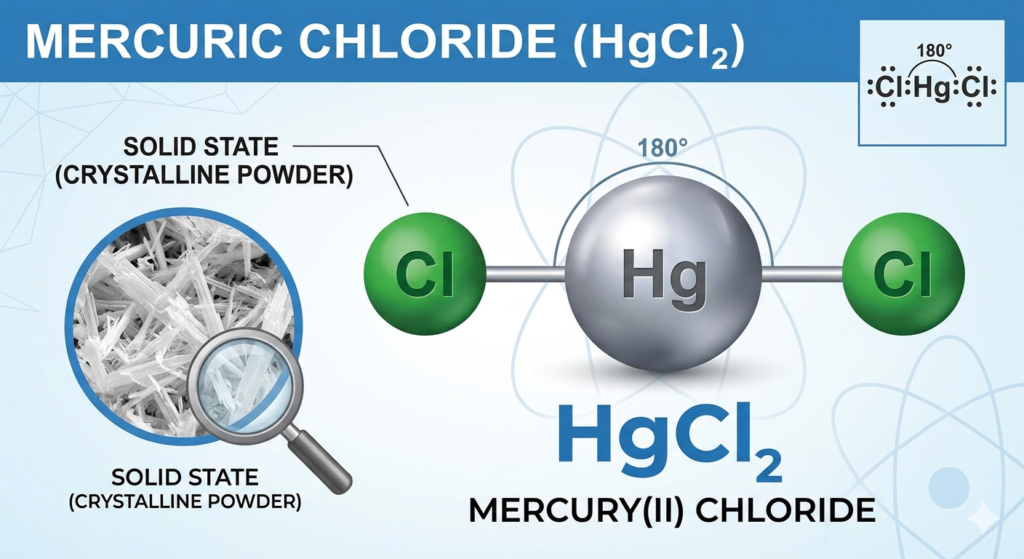
2. Chemical Structure and Properties
- Molecular Formula: HgCl₂
- Molar Mass: 271.50 g/mol
- IUPAC Name: Mercury(II) chloride
- Structure: Linear molecule (Cl–Hg–Cl)
- Oxidation State: +2 for mercury
Key Chemical Features
- Polarity: Highly polar, resulting in strong interactions with water and polar solvents.
- Solubility: Dissolves readily in water, alcohol, and ether.
- Reactivity: Acts as a strong oxidizing and chlorinating agent, making it useful in organic and inorganic chemistry.
3. Physical Characteristics
- Appearance: White, odorless, crystalline solid
- Density: 5.43 g/cm³
- Melting Point: 277°C
- Boiling Point: 302°C (decomposes upon boiling)
- Solubility in Water: 6.0 g/100 mL at 20°C
- Vapor Pressure: Low, but can sublime at elevated temperatures
Note: Direct contact or inhalation is extremely dangerous due to the compound’s toxicity.
4. Historical Background
Mercuric chloride has been known since ancient times, with early alchemists using it for various mystical and practical purposes. Its alternative name, “corrosive sublimate,” stems from its ability to change directly from solid to vapor (sublimation) when heated.
Key Historical Points
- Middle Ages: Used as a pigment, preservative, and in alchemical experiments.
- 18th–19th Centuries: Employed as a disinfectant, antiseptic, and even a treatment for syphilis (with disastrous health effects).
- 20th Century Onwards: Widespread use replaced by safer alternatives in medicine and consumer products, but retained in specialized industrial and research settings.
5. Synthesis and Industrial Production
Main Production Methods
- Direct Chlorination:
Hg (liquid mercury) + Cl₂ (chlorine gas) → HgCl₂ - Double Displacement:
HgSO₄ + 2NaCl → HgCl₂ + Na₂SO₄
These methods yield mercuric chloride of high purity for laboratory and industrial supply.
6. Laboratory and Industrial Uses
Laboratory Applications
- Analytical Chemistry:
- Reagent for detecting alkaloids, proteins, and other compounds.
- Used in gravimetric and titrimetric analyses.
- Organic Synthesis:
- Chlorinating agent and catalyst in forming organomercury compounds.
- Histology:
- Fixative for preserving and staining tissues for microscopy.
- Reference Material:
- Standard for calibration and quality control.
Industrial Applications
- Catalyst in Manufacturing:
- Essential in producing vinyl chloride, a precursor to PVC plastics.
- Biocide:
- Used in water treatment and as a fungicide/preservative (now rare).
- Photography:
- Historical use in photographic intensifiers.
Note: Due to its hazardous nature, many historical uses have been discontinued or banned.
7. Historical and Obsolete Medical Applications
Mercuric chloride was once common in medicine, but all such uses are now obsolete for safety reasons.
- Antiseptic and Disinfectant:
Applied to wounds and surgical tools. - Syphilis Treatment:
Used before penicillin, despite severe side effects. - Laxative and Diuretic:
Included in some 19th-century remedies. - Preservative:
Used in vaccines and anatomical specimens.
Warning: Use in human or veterinary medicine is now strictly prohibited.
8. Safety, Toxicity, and Exposure Risks
Mercuric chloride is extremely toxic and must be handled with the highest caution.
Routes of Exposure
- Ingestion: Acute poisoning, severe organ damage, potentially fatal.
- Inhalation: Lung irritation, systemic toxicity.
- Skin Contact: Absorbed through the skin; even small amounts can cause poisoning.
- Chronic Exposure: Neurological and renal damage, memory loss, tremors, irritability (“mad as a hatter” syndrome).
Symptoms of Poisoning
- Abdominal pain, vomiting, diarrhea
- Renal failure
- Tremors, confusion, irritability
- Death (in severe or untreated cases)
First Aid
- Ingestion: Seek immediate medical attention; do NOT induce vomiting.
- Skin Contact: Remove contaminated clothing, wash skin thoroughly.
- Inhalation: Move to fresh air, seek emergency care.
9. Environmental Impact
Mercuric chloride is an environmental hazard:
- Bioaccumulation: Mercury compounds persist in the environment and accumulate in living organisms.
- Water & Soil Contamination: Improper disposal leads to long-term ecological harm.
- Air Pollution: Sublimation at high temperatures may release toxic vapors.
Strict environmental regulations govern its disposal and spill response.
10. Handling, Storage, and Disposal
Handling
- Always wear gloves, goggles, and lab coats.
- Work in a fume hood or well-ventilated space.
- Avoid all direct contact and inhalation of dust or vapors.
Storage
- Store in tightly sealed, clearly labeled containers.
- Keep in cool, dry, secure locations away from incompatible substances (e.g., ammonia, acetylene).
- Restrict access to trained and authorized personnel.
Disposal
- Never dispose of down the drain or in general waste.
- Collect all waste for hazardous disposal by licensed contractors.
- Follow all South African environmental and hazardous waste regulations.
11. Regulatory Status in South Africa
Mercuric chloride is a hazardous substance under South African law:
- Purchase & Use: Only licensed organizations and professionals may buy or use.
- Transport: Must comply with local and international hazardous goods regulations.
- Documentation: Suppliers must provide SDS, COA, and necessary permits.
- Workplace: Employers must provide training, risk assessments, and emergency procedures.
Brand Chemical Supplies guarantees all regulatory compliance and provides full documentation and support.
12. How to Purchase Safely from Brand Chemical Supplies
Steps for Safe Procurement
- Determine Your Needs:
- Grade, quantity, intended application.
- Contact Brand Chemical Supplies:
- Phone, email, or web inquiry.
- Provide Required Documentation:
- Business license, relevant permits, intended use.
- Receive a Formal Quotation:
- Includes price, VAT, delivery, and compliance documents.
- Arrange for Secure Delivery:
- Ensure proper facilities for receipt and storage.
- Review Safety Data:
- Train staff and implement risk controls.
13. Frequently Asked Questions
Q: What is the formula for mercuric chloride?
A: HgCl₂
Q: What is the minimum order quantity?
A: 5 kg (standard for most suppliers due to packaging and regulation)
Q: Can individuals buy mercuric chloride?
A: No, only licensed professionals and organizations.
Q: Is mercuric chloride still used in medicine?
A: No, all medical uses are obsolete due to toxicity.
Q: How is mercuric chloride delivered?
A: Secure packaging, compliant courier delivery, tracking provided.
Q: What documentation is included?
A: Safety Data Sheet (SDS), Certificate of Analysis (COA), permits.
Q: What are the main hazards?
A: Acute and chronic mercury poisoning, severe organ damage, environmental harm.
Q: How do I dispose of mercuric chloride waste?
A: As hazardous waste, via certified contractors and compliant with local laws.
14. References and Further Reading
- World Health Organization – Mercury and Health
- South African Chemical Regulations
- PubChem – Mercury(II) chloride
- [Brand Chemical Supplies SDS Portal] (Insert link if available)
15. Conclusion
Mercuric chloride (HgCl₂) is a potent, versatile, but extremely hazardous chemical. Its role in South Africa’s laboratories and industries is critical but demands strict adherence to safety and legal standards. Always source from a reputable, licensed supplier like Brand Chemical Supplies to ensure quality, compliance, and environmental responsibility.
Contact Brand Chemical Supplies for up-to-date pricing, technical support, and secure supply of mercuric chloride and other regulated chemicals across South Africa.
Brand Chemical Supplies – Your Trusted Source for Safe, Compliant Laboratory and Industrial Chemicals.