Mercuric Chloride in Medical Research: Historical Legacy, Modern Applications, and Safety
Introduction
Mercuric chloride (HgCl₂), also known as corrosive sublimate, is a chemical compound with a complex and storied history in medical research and healthcare. Once celebrated for its potent antimicrobial properties and diverse applications in medicine, mercuric chloride has, over time, become recognized for its significant toxicity and environmental risks. Despite these concerns, its unique chemical characteristics continue to make it a valuable tool in select areas of modern medical research, histology, and analytical science. This comprehensive guide explores the evolution of mercuric chloride’s role in medical research, detailing its mechanisms, applications, regulatory landscape, safety requirements, case studies, and future outlook.
The Historical Context of Mercuric Chloride in Medicine
Early Medical Use
Mercuric chloride’s use in medicine dates back to the 18th and 19th centuries. Known then as “corrosive sublimate” due to its appearance and properties, it was widely employed for its broad-spectrum antimicrobial effects.
Key Historical Applications:
- Topical antiseptic: Used to clean wounds and prevent infection before the advent of antibiotics.
- Syphilis treatment: Administered orally or by injection, despite severe toxicity and a small margin of safety.
- Disinfectant: Applied to surgical instruments, medical facilities, and even clothing to control the spread of disease.
- Preservative: Used to preserve biological specimens, anatomical samples, and even some pharmaceutical products.
Rise and Decline
Mercuric chloride’s effectiveness as an antimicrobial and preservative led to its widespread adoption. However, its high toxicity quickly became apparent. Reports of mercury poisoning, renal failure, and neurological symptoms led to increased scrutiny. By the early 20th century, the introduction of safer antiseptics and antibiotics, such as penicillin, drastically reduced its medical use.
Chemistry and Mechanism of Action
Chemical Properties
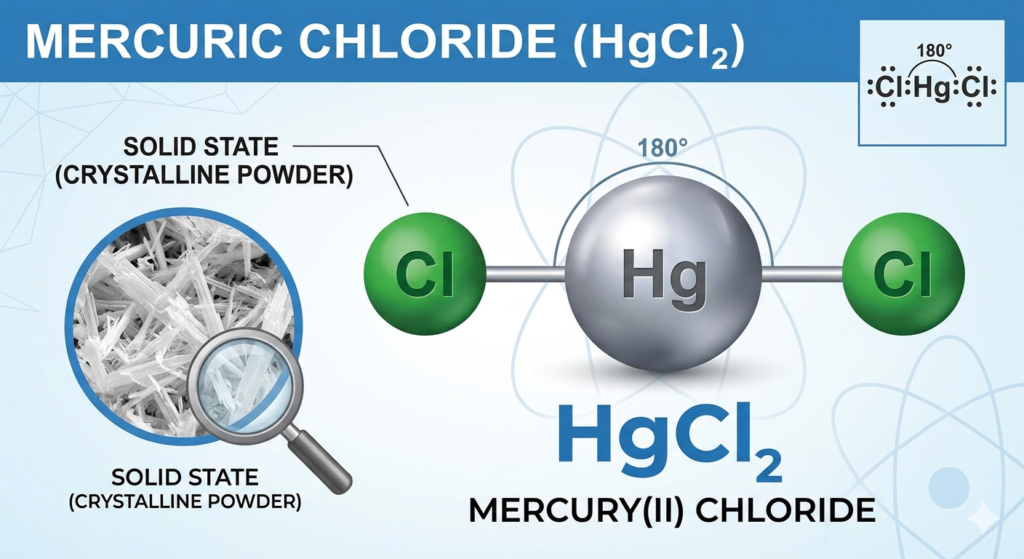
- Formula: HgCl₂
- Appearance: White, odorless crystalline substance
- Solubility: Highly soluble in water and organic solvents
- Reactivity: Acts as a strong oxidizing and protein-denaturing agent
Mechanism in Biological Systems
Mercuric chloride is highly reactive with thiol (sulfhydryl) groups in proteins and enzymes. This disrupts their normal functions, resulting in:
- Antimicrobial action: Rapidly kills bacteria, fungi, and some viruses by denaturing essential cellular proteins.
- Cellular toxicity: Damages mammalian cells in the same way, leading to the compound’s well-documented human toxicity.
Modern Applications in Medical Research
While no longer used therapeutically, mercuric chloride retains important roles in research laboratories, diagnostics, and pharmaceutical testing due to its unique properties.
1. Histology and Pathology
Tissue Fixation
Mercuric chloride is a core component of several classical fixatives, most notably Zenker’s fixative. Its ability to cross-link proteins and preserve fine cellular detail makes it invaluable for:
- Routine histological staining
- Microscopic examination of tissue architecture
- Immunohistochemistry (in specific protocols)
Advantages:
- Superior nuclear and cytoplasmic detail
- Consistent tissue preservation
Disadvantages:
- Mercury waste disposal issues
- Health hazards for laboratory workers
2. Biochemistry and Protein Research
Mercuric chloride is used in:
- Protein precipitation: Selectively denaturing and precipitating proteins to isolate specific fractions for study.
- Enzyme inhibition studies: As a potent inhibitor, HgCl₂ helps researchers understand enzyme mechanisms and design inhibitors for drugs.
- Standard in analytical chemistry: Used as a reference for mercury detection and quantification in pharmaceutical analysis.
3. Microbiology
Selective Media Preparation
- Inhibition of unwanted flora: Mercuric chloride is sometimes added to culture media to suppress certain bacteria or fungi, allowing for the isolation of specific pathogens.
- Antimicrobial susceptibility testing: Used in research to measure the effects of mercury and study resistance mechanisms.
4. Pharmaceutical Quality Assurance
Trace Analysis
- Ensuring pharmaceutical products are free from mercury contamination
- Used as a calibration standard for mercury detection in drugs, vaccines, and raw materials
5. Toxicology and Environmental Medicine
- Mercury toxicity studies: Used to induce and model mercury poisoning in laboratory animals, advancing understanding of mercury’s effects on human health.
- Chelation therapy research: Helps scientists evaluate new chelating agents for treating heavy metal poisoning.
Safety Protocols and Laboratory Handling
Given its high toxicity, all use of mercuric chloride in medical research is governed by strict safety protocols.
Personal Protective Equipment (PPE)
- Gloves: Impermeable to mercury compounds
- Lab coats and gowns
- Eye protection: Goggles or face shields
- Respiratory protection: When working with powders or vapors
Engineering Controls
- Fume hoods: Required for all manipulations to prevent inhalation of dust or vapors
- Mercury spill kits: Available and staff trained in their use
- Segregated storage: Away from incompatible chemicals (e.g., ammonia, strong bases)
Waste Management
- Waste labeled as hazardous: All mercury-containing waste is collected separately
- Certified disposal: Must be disposed of via licensed hazardous waste handlers
- Environmental monitoring: Regular environmental audits for mercury contamination
Training and Documentation
- Safety Data Sheets (SDS): Must be reviewed and accessible to all personnel
- Staff training: Regular instruction on handling, spill response, and emergency procedures
- Compliance audits: Regular reviews to ensure adherence to regulations
Regulatory and Ethical Considerations
International and National Regulations
Mercuric chloride is classified as a hazardous substance by many global agencies, including:
- REACH (EU): Registration, Evaluation, Authorisation, and Restriction of Chemicals
- OSHA and EPA (USA): Occupational and environmental safety standards
- Minamata Convention: Global treaty to reduce mercury pollution
Procurement and Permitting
- Only licensed institutions may purchase mercuric chloride for research use
- Permits and documentation required for acquisition, storage, and disposal
Ethical Considerations
- Minimizing use: Substitute less hazardous reagents where possible
- Staff safety: Adherence to best practices to protect personnel
- Environmental stewardship: Preventing environmental contamination and community exposure
Case Studies: Mercuric Chloride in Modern Medical Research
Case Study 1: Histopathology Laboratory
A major teaching hospital continues to employ Zenker’s fixative (containing mercuric chloride) in specific diagnostic protocols where superior tissue and nuclear detail are required. The laboratory has strict safety, waste, and documentation policies, and invests in regular staff training and environmental monitoring.
Case Study 2: Pharmaceutical Analytical Lab
A pharmaceutical quality control laboratory uses mercuric chloride AR as a calibration standard for mercury detection. Precise trace analysis ensures that finished products are free from mercury contamination, thus protecting patient safety and regulatory compliance.
Case Study 3: Toxicology Research Center
Researchers at a toxicology center use mercuric chloride to create animal models of acute and chronic mercury poisoning. This research informs new treatments and chelation therapies for mercury exposure, as well as public health recommendations.
Advances and Alternatives: The Future of Mercuric Chloride in Research
Shift to Safer Reagents
Modern science increasingly favors non-mercury alternatives for tissue fixation, antimicrobial testing, and protein research. New reagents and protocols offer similar or improved performance with reduced risks.
Examples:
- Formalin: Widely used for tissue fixation in place of mercury-based fixatives
- Thimerosal-free vaccines: Mercury-containing preservatives have been replaced in most countries
Green Chemistry and Sustainability
- Reduced mercury use: Global initiatives (such as the Minamata Convention) encourage reduced reliance on mercury in all sectors, including research
- Improved waste handling: Technologies for mercury capture and remediation are advancing
Frequently Asked Questions (FAQ)
Q: Is mercuric chloride still used in clinical medicine?
A: No, direct clinical use is obsolete due to toxicity. Its use is now limited to laboratory research and analytical contexts.
Q: What are the main safety concerns with mercuric chloride in research?
A: Acute and chronic toxicity, environmental persistence, and strict handling/disposal requirements.
Q: Can researchers substitute safer chemicals for mercuric chloride?
A: Yes, many protocols now use safer alternatives, but in some cases, the unique properties of mercuric chloride remain essential.
Q: How is mercury waste managed in medical labs?
A: All mercury-contaminated waste is collected, labeled, and disposed of through certified hazardous waste handlers, with strict documentation.
Q: Who can purchase mercuric chloride for research purposes?
A: Only licensed institutions with appropriate permits and regulatory compliance.
Sourcing: Choosing a Reputable Supplier
When mercuric chloride AR is required, sourcing from a reputable supplier ensures:
- Certified AR grade: Meets analytical reagent standards for purity
- Full documentation: Certificates of Analysis (COA) and Safety Data Sheets (SDS) provided
- Compliance support: Assistance with regulatory and shipping requirements
Brand Chemical Supplies is a leading supplier, providing high-purity mercuric chloride for research, with global shipping and compliance support.
Conclusion
Mercuric chloride’s journey in medical research is a reflection of advances in science, medicine, and safety awareness. While its therapeutic use has been replaced by safer alternatives, its unique chemical properties maintain its relevance in a select range of research, analytical, and diagnostic applications. Modern use is characterized by strict regulatory oversight, comprehensive safety measures, and a growing emphasis on environmental stewardship. As science continues to evolve, new technologies and safer substitutes will further minimize dependence on hazardous compounds like mercuric chloride, ensuring that medical research remains both innovative and responsible.
Keywords: Mercuric chloride medical research, AR grade mercuric chloride, laboratory fixative, histology, protein precipitation, mercury toxicity, pharmaceutical analysis, hazardous chemical handling, safety protocols, Brand Chemical Supplies, regulated chemical purchase, environmental compliance, mercury waste management, medical laboratory research.
For certified mercuric chloride AR and compliance support, contact Brand Chemical Supplies or consult the latest Safety Data Sheet (SDS) for guidance on safe handling and disposal.